Lose weight naturally & sustainably with THINNR at Alani Skin MD - a doctor-supervised, all natural weight loss program.
At Alani Skin MD in Phoenix, AZ, we offer the THINNR Weight Loss Program, a doctor-supervised, all-natural approach to sustainable weight loss.
Unlike restrictive fad diets or medications with side effects, THINNR works with your body’s metabolism to promote healthy, long-term fat loss without prescriptions or injections.
Fill out the form and our team will be in touch within 24 hours.
Your information is 100% confidential. We never share your data.
THINNR is a 6-week, provider-guided program that helps you lose weight naturally while maintaining optimal health. It works through a 4-phase approach designed to optimize your metabolism, reduce inflammation, and promote lasting weight loss results.
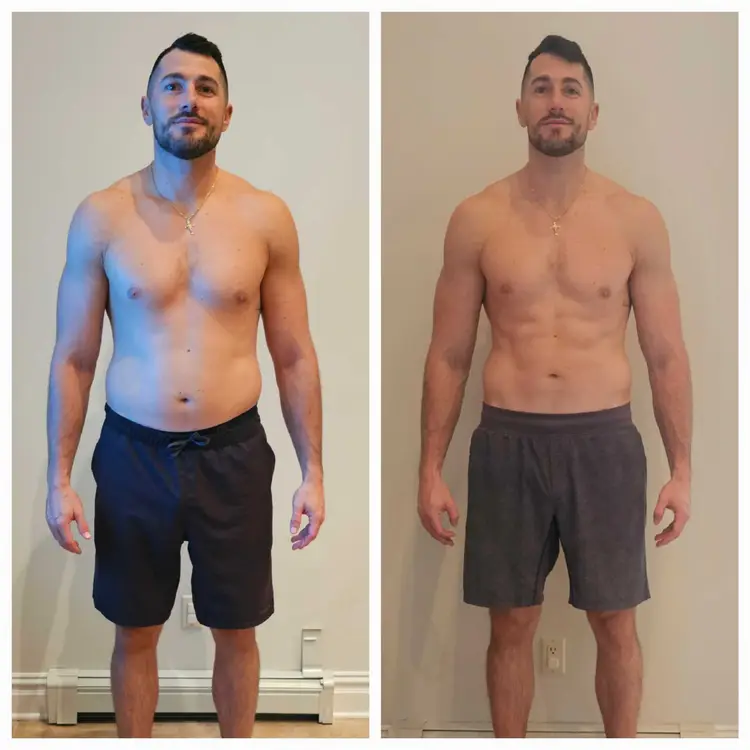
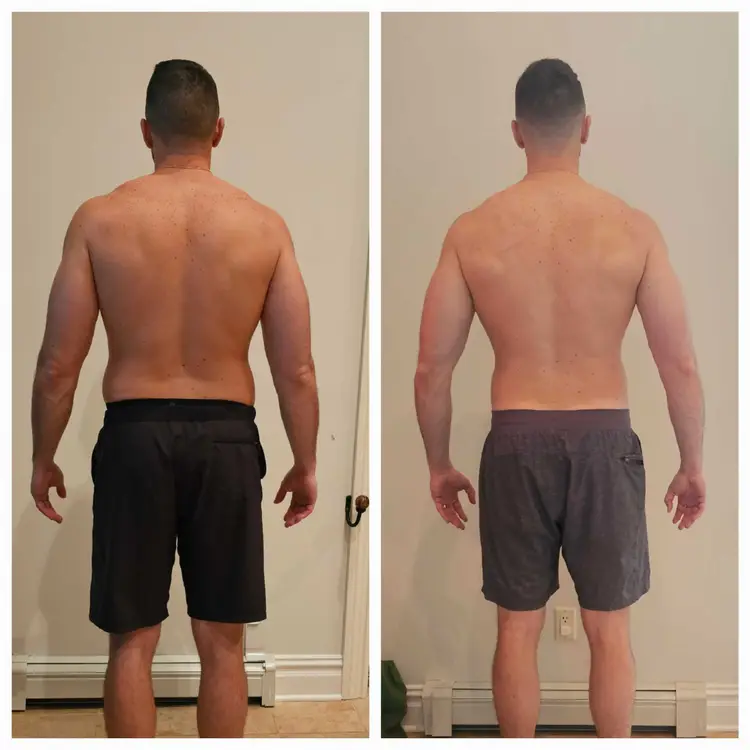
THINNR patients commonly lose 1–3 lbs per day the first week and 0.5–0.70 lbs daily on the body.
The THINNR App provides community support and a personal guide to help you stay on course.
At Alani Skin MD, we go beyond just providing the THINNR program.
Our comprehensive, medically guided approach ensures you achieve maximum results safely and effectively.
Tailored to your unique health needs — not a one-size-fits-all approach.
Monitor progress & adjust for optimal results throughout the program.
Stay connected with your coach, track progress, and access resources anytime.
Sustainable meal plans designed for real results.
We’re with you every step of the way — you’ll never feel alone on this journey.
With our expert guidance, you won’t just lose weight — you’ll transform your health and lifestyle for the long run.
AVERAGE WEIGHT LOSS
1–3 lbs / day
First week results vary by individual
PATIENT SATISFACTION
97%
Would recommend THINNR to a friend
PROGRAM COMPLETION RATE
91%
Patients complete their full 42-day program
Everything you need to know before getting started.
The Thinnr Weight Loss Protocol harnesses a 21-day approach to weight loss while maintaining the 4 essential keys to sustainable long-term weight loss maintenance. The Thinnr program plans are the Thinnr Homeopathic OTC Medication and the Thinnr Weight Loss & Weight Maintenance Nutrition Strategy. The Thinnr homeopathic medication is an all-natural, FDA-registered homeopathic OTC medicine that promotes healthy weight management by optimizing metabolism, lower stress levels, reduced hunger and cravings, balanced diet, and improved elimination (part of a comprehensive weight management plan).
All products in THINNR’s product portfolio are manufactured in US-based, FDA-registered, cGMP-certified, and state-licensed facilities that the FDA routinely inspects. Additionally, all THINNR medications and supplements are put through extensive post-production quantitative testing to ensure product specification, potency, and safety are met before being released and made available for sale.
All THINNR products are manufactured in accordance with Good Manufacturing Practices and the Code of Federal Regulations (CFRs).
While weight loss will vary by person, many people have lost and could lose between 20-35 pounds on the 42-day THINNR protocol. Everyone is different. It depends on many factors, including how much excess weight you have, how much weight needs to be lost, genetics, and an individual’s follow-through and adherence to any weight loss THINNR protocol they are performing.
While it is common to feel mild psychological hunger during the first few days, this sensation stops after 4-6 days.
Many times, there will be a large amount of weight loss in the first week, then a gradual slowing. This does not mean that your weight loss has stopped. After a period of time, patients will notice another large weight loss. Typically, inches are continuously being lost while you are on the THINNR protocol.
Given the homeopathic nature of these ingredients, significant interactions with conventional drugs are not highly likely. Should you have any specific concerns, please consult your Thinnr provider.
None of the listed ingredients are explicitly known to cause changes in the menstrual cycle as a direct effect. Likewise, THINNR will not affect your ability to become pregnant, nor will it increase your chances of getting pregnant. It is best to avoid starting the THINNR protocol if you are within 5-6 days of your menstrual cycle. If you are currently taking fertility medications, we advise against starting the THINNR protocol. Occasionally, a woman may experience spotting or an unexpected menstrual cycle. This is due to the fact that estrogen is stored in fat cells, and as the fat cells are used to provide energy to the body, the stored estrogen is released into the bloodstream.
A woman is prohibited from participating in the THINNR protocol if she is pregnant. If you become pregnant during the THINNR protocol, stop taking THINNR immediately and consult your physician.
Never skip any of your meals. It's important to eat all your allotted food. It is especially important that you consume the full portions of protein. Additionally, do not move portions of food from one meal to the next.
Exercise is not required to see success with the Thinnr program. Refer to the exercise section of this book for more details on exercise.
Some people may experience light headaches during the first week. There are 2 main reasons. The first is that your body is withdrawing from simple carbohydrate consumption. The other reason is related to your body going through a detoxification process. If you experience a headache, please ensure you are drinking the correct amount of water and consider taking a high-quality electrolyte supplement or receiving an IV hydration treatment with electrolytes.
No. After 42 days, you need to enter the Metabolic Normalization phase for a minimum of 3 weeks.
It’s strongly recommended that you not consume alcohol while on THINNR.
Join hundreds of patients who have transformed their health with the THINNR program at Alan Skin MD.
How It Works
Why THINNR
Book Consultation
(602) 464-9906
© 2026 THINNR · Alani Skin MD · Phoenix, AZ. All rights reserved. These statements have not been evaluated by the Food and Drug Administration.